Uveal melanoma (UM) is a malignancy arising from the vascular tunic and also the most common malignant intraocular tumor. UM is highly malignant and easy to metastasize. About one-half of the UM patients arediagnosed with liver metastases, andthey only have a limited survival after being diagnosed with metastases. The median progression-free survival and the overall survival of patients with liver metastases are 3.3 and 10.2 months, respectively. Unlike melanoma in the skin, mucosa and other parts of the body, UM is insensitive to chemotherapy, radiotherapy and immunotherapy. In a word, these conventional therapies have very limited efficacy in improving the survival rate and survival length. UM is usually diagnosed at the advanced stage due to its hidden onset and lack of knowledge about this disease.UMpatientsreaching anadvanced stage and being in a critical condition account for a large proportion.Identifyingnovel therapeutic targets in UM and reducing the mortality of UM are clinical challenges awaiting solution.
 Fig. 1: Title page Recently, the teams led by Academician Fan Xianqun from Shanghai Jiao Tong University School of Medicine and by Professor Lei Ming, the executive dean of the Institute of Precision Medicine at Shanghai Jiao Tong University School of Medicine, have worked together to reveal the mechanism of allosteric activation of key enzymes involved in gluconeogenesis in UM. Their research has also clarified how acetyl-CoA regulates pyruvate carboxylase (PC), a key problem that was once unresolved over the years. Their findings have been publishedin Molecular Cell, a sub journal ofCell. (Fig. 1)
 Academician of CAE, Fan Xianqun. According to the preliminary study conducted by the department of ophthalmology at the Shanghai Ninth People's Hospital affiliated with the Shanghai Jiao Tong University School of Medicine, themetabolic reprogramming is one malignant feature of UM. Tumor cells adapt to the hypoxic tumor microenvironment via anaerobic glycolysis, glutamine metabolism, and energy supply by non-principal sources of energy. These mechanisms are crucialto meetthe material and energy demands of tumor cells during rapid proliferation (Yu et al., Genome Biol, 2021). Another study has shown that the abnormal activation of gluconeogenesis is closely related to liver metastases in UM and therefore acts as an ideal therapeutic target for metastastic UM (Nat Rev Cancer. 2013). PC is identified as a key enzyme involved in the catalytic carboxylation of pyruvic acid to produce oxaloacetic acid. This catalytic process is also the first rate-limiting step in gluconeogenesis, where acetyl-CoA acts as an important metabolite of allosteric activation. However, the role of allosteric activation of PC mediated by acetyl-CoA in the pathogenesis of UM remains unclear.
 Professor Lei Ming doing scientific experiments. Addressing this critical scientific issue, Dr. Chai Peiwei from the department of ophthalmology at the Shanghai Ninth People's Hospital affiliated with the Shanghai Jiao Tong University School of Medicine and the research fellow Lan Pengfei at the Institute of Precision Medicine at Shanghai Jiao Tong University School of Medicine introduced four-point mutations in human PC, asinstructedby Academician Fan Xianqun and Professor Lei Ming. This resulted in the destruction of the binding domain between acetyl-CoA and PC,which promotedthe self-activation of PC. The research team found that the expression of self-activated PC significantly inhibited the growth and colony formation of UM cells. An overexpression of self-activated PC led to a significant increase in mitochondrial oxaloacetate and a reduction in acetyl-CoA. The above result indicated that an elevated activity of self-activated PC consumed more pyruvate for oxaloacetate production so less pyruvate could be converted to acetyl-CoA by pyruvate dehydrogenase. Accompanying decreased acetyl-CoA, the researchers also observed a reduced level of citrate in self-activated PC mutant -expressing cells, consistent with the fact that most acetyl-CoA in mitochondria is consumed for citrate synthesis. The above results demonstrate that the functional PC sensitive to acetyl-CoA regulation is essential for metabolic homeostasis and oxidative respiration in PC-independent cells. (Fig. 2)
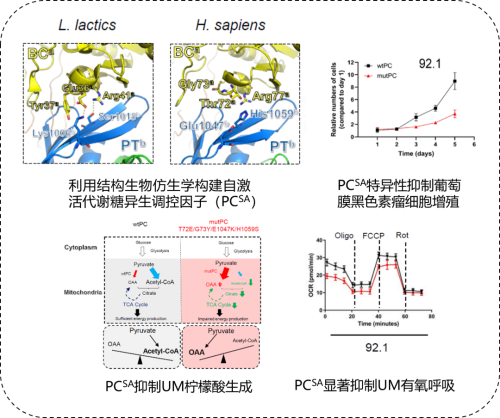 Fig. 2 So the next question is how acetyl-CoA induces the stability of biotin carboxylase (BC) domain in PC complexes. The researchers captured PC structures in the presence of acetyl-CoA at a series of concentrations and purified BC and carboxyltransferase (CT) domains as separate proteins. Comparative analysis of these structures revealed a striking correlation between the stability of the BC domains and the acetyl-CoA concentration. Further cryo-EM analysis demonstrated that acetyl-CoA also induces the stabilization of the BC domains in the BC-(PT-CT-BCCP) complex, resulting in a conformation that is indistinguishable to the native PC complex. Taken together, these results suggest that acetyl-CoA functions as a mediator for the stable association between BC and PT-CT-BCCP, which helps shape a more stable architecture of BC and PT-CT-BCCP (Fig. 3). This is in perfect alignment with previous findings that acetyl-CoA decreases the activation entropy and induces a much more ordered thermodynamic configuration during PC catalytic turnover, which could not be explained by previous crystallographic structural studies.
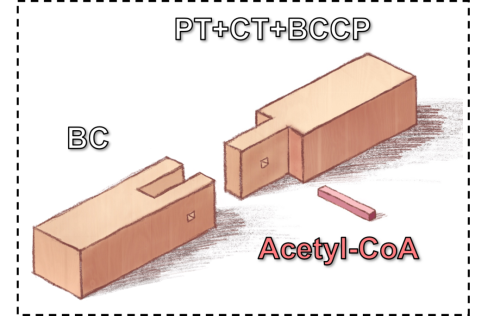 Fig. 3 Acetyl-CoA functions as a mediator for the stable association between BC and PT-CT-BCCP PC is an important metabolism-regulating enzyme and plays key roles in insulin secretion, adipogenesis, and neurotransmitter biosynthesis. It is also an extensively studied therapeutic target in obesity, diabetes, viral infections and cancers. The allosteric effect of PCis crucial formetabolic reprogramming in many diseases.Mapping the structure of the PC/acetyl-CoA complexwill inform the research and development of medications against obesity, diabetes, viral infections and cancers. A brief introduction to the team leaders Professor Fan Xianqun, Academician of Chinese Academy of Engineering, appointed as Changjiang Distinguished Professor, an ophthalmology expert, and chair professor of Shanghai Jiao Tong University. He is currently Vice President of Shanghai Jiao Tong University, Chancellor of Shanghai Jiao Tong University School of Medicine, Dean of China Hospital Development Institute, director of Shanghai Key Laboratory of Orbital Diseases and Ocular Oncology, and ophthalmic leader at Shanghai Ninth People's Hospital affiliated to Shanghai Jiao Tong University School of Medicine. He serves concurrently as the President of Asia-Pacific Society of Ocular Oncology & Pathology, the fifth president of the Asia Pacific Society of Ophthalmic Plastic and Reconstructive Surgery, member of the Academia Ophthalmologica Internationalis, Fellow of the Royal College of Ophthalmologists, and Fellowship of the Royal College of Surgeons of Edinburgh. He won the Second prize of National Science and Technology Progress Award twice (as the first accomplisher), Ho Leung Ho Lee Science and Technology Progress Award, the first Annual Innovator Award of Asia-Pacific Academy of Ophthalmology, and the De Ocampo Lecture, the highest award to ophthalmologists from the Asia-Pacific region. Lei Ming, research fellow and doctoral supervisor, now working as the executive dean of the Institute of Precision Medicine at Shanghai Jiao Tong University School of Medicine. He has committed himself to the research of the structure and function of biological macromolecules and their roles in cancers and aging. He has published over 60 articles in the top international academic journals, includingCell,Nature andScience. |

