Recently, a landmark study led by Academician Xia Qiang, President of Renji Hospital affiliated withShanghai Jiao Tong University School of Medicine (SJTUSM), was published in the top-tier international journalNature Medicine. This study represents the world’s first Phase I clinical trial using liver-targeted in vivo base editing to treat heterozygous familial hypercholesterolemia (HeFH). The research demonstrated, for the first time in humans, both the safety and efficacy of this gene therapy, offering a novel approach to relieving patients of lifelong medication burden.
HeFH is a common inherited metabolic liver disorder caused by mutations in genes such as the low-density lipoprotein receptor (LDLR),leading to congenitaldefects in hepatic cholesterol metabolism. From birth, affected individuals exhibit persistently elevated low-density lipoprotein cholesterol (LDL-C), placing them at extremely high risk for early-onset coronary heart disease, myocardial infarction, and other atherosclerotic cardiovascular diseases. Prior to this study, treatment was limited to lifelong regular medication, posing significant challenges in adherence and treatment burden.
To address this unmet clinical need, the team led by Academician Xia, including the liver surgery groups led by Wan Ping and Yang Taihua, developed a novel in vivo gene editing strategy with support from the Shanghai Key Laboratory of Precision Gene Editing and Clinical Translational Technology, theSino-German Gene and Cell Therapy Research Center, Shanghai Jiao Tong University, andYolTech Therapeutics. This approach uses a high-precision adenine base editor to selectively silence the hepatic proprotein convertase subtilisin/kexin type 9 (PCSK9) gene. Delivery is achieved via a dual-pathway liver-targeted system using N-acetylgalactosamine (GalNAc)-modified lipid nanoparticles (LNPs). This design allows selective editing of hepatocytes while preserving LDLR function, making it particularly suitable for HeFH patients with LDLR defects.
The Phase I trial enrolled six patients across three dosing groups (0.2, 0.4, and 0.6 mg/kg). Both efficacy and safety were rigorously assessed. In the high-dose 0.6 mg/kg group, after 24 weeks of treatment, serum PCSK9 levels decreased by an average of 74.4%; Serum LDL-C levels decreased by an average of 52.3%; Lipid-lowering effects were sustained and stable. Safety outcomes were favorable: no grade≥3 adverse events occurred, and no participants withdrew due to adverse effects. The most common mild infusion-related reactions were fever and myalgia, which resolved spontaneously within 24 hours. Transient elevationsin liver transaminases were observed in some patients, which returned to normal shortly thereafter. Comprehensive off-target analysis at boththe DNA and RNA levels detected no unintended edits, confirming the therapy’s precision.
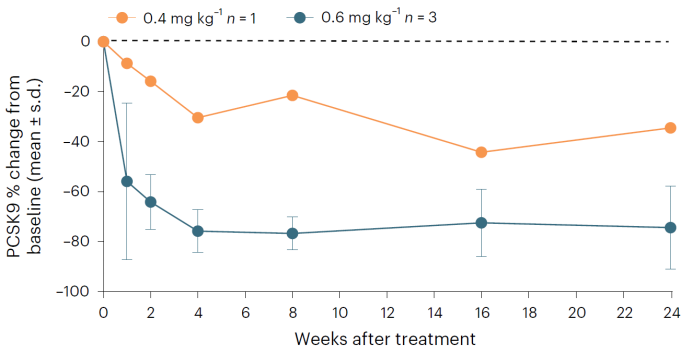
Post-Gene Therapy Serum PCSK9 Level Changes
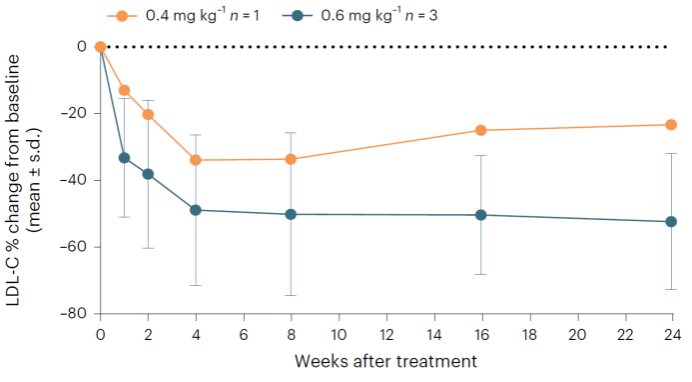
Post-Gene Therapy Serum LDL-C Level Changes
Co-first authors of the study include Dr. Wan Ping, Associate Chief Physician, Liver Surgery, Renji Hospital, postdoctoral fellows Tang Siyuan, Lin Dongni, and doctoral student Long Mei, as well as Professor Lu Yuming, Shanghai Jiao Tong University. Academician Xia Qiang of the Chinese Academy of Engineering and President of Renji Hospital, Yang Taihua, resident physician and associate researcher in the Department of Liver Surgery, Professor Wu Yuxuan of East China Normal University, and Dr. Wang Zijun of YolTech Therapeutics served as co-corresponding authors of the study.The study was supported by major national science and technology projects for chronic diseases, the National Natural Science Foundation of China (Outstanding Research Group, Category A), and other funding sources.
This study represents a significant breakthrough in the treatment of heterozygous familial hypercholesterolemia. Witha single intravenous infusion of the gene-editing therapy, patients can achieve long-term, stable lipid reduction. The therapeutic effect is comparable to that of existing PCSK9 monoclonal antibodies, yet it eliminates the need for lifelong regular medication, fundamentally addressing the adherence challenges associated with conventional treatments.
Xia Qiang noted that the lifelong medication required for patients with familial hypercholesterolemia imposes a substantial treatment burden. This study is the first to demonstrate that a single intravenous infusion of base-editing therapy can achieve durable lipid lowering, offering new hope for reducing patients’ medication burden and enabling long-term disease control. According to the research team, participants in the clinical trial will undergo long-term follow-up for up to 15 years to continuously monitor treatment outcomes. Meanwhile, the team will further optimize the lipid nanoparticle (LNP) formulation to enhance the therapy’s clinical benefits and advance this innovative treatment toward more mature clinical applications.

