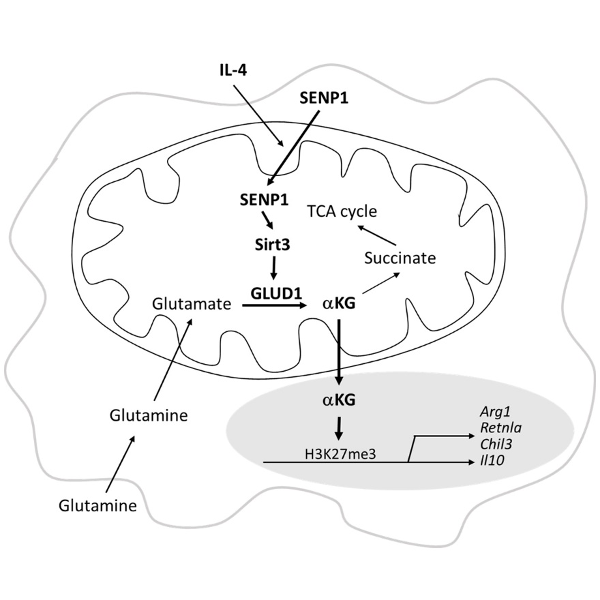
The article SENP1-Sirt3 signaling promotes α-ketoglutarate production during M2 macrophage polarization was published in Cell Reports by Professor Jinke Cheng Lab of Basic Medical Sciences on 12 April 2022, reported a new mechanism of SENP1-Sirt3 signaling promoting α-ketoglutarate production during M2 macrophage polarization through regulating glutamine metabolism.
Abstract
The metabolic program is altered during macrophage activation and influences macrophage polarization. Glutaminolysis promotes accumulation of α-ketoglutarate (αKG), leading to Jumonji domain-containing protein D3 (Jmjd3)-dependent demethylation at H3K27me3 during M2 polarization of macrophages. However, it remains unclear how αKG accumulation is regulated during M2 polarization of macrophages. This study shows that SENP1-Sirt3 signaling controls glutaminolysis, leading to αKG accumulation during IL-4-stimulated M2 polarization. Activation of the SENP1-Sirt3 axis augments M2 macrophage polarization through the accumulation of αKG via glutaminolysis. We also identify glutamate dehydrogenase 1 (GLUD1) as an acetylated protein in mitochondria. The SENP1-Sirt3 axis deacetylates GLUD1 and increases its activity in glutaminolysis to promote αKG production, leading to M2 polarization of macrophages. Therefore, SENP1-Sirt3 signaling plays a critical role in αKG accumulation via glutaminolysis to promote M2 polarization.
Link: https://www.cell.com/fulltext/S0092-8674(07)01143-9
About Lab
Laboratory of Cell Signal Transduction
The lab targets protein SUMOylation, especially SENPs-mediated deSUMOylation and its regulation mechanism of cell signal transduction, as well as the significance in body development and disease.
The research interests include:
1) Protein SUMOylation on cellular metabolism: To study the molecular mechanism of mitochondrial protein SUMOylation on mitochondrial metabolism and its effect on cellular function.
2) Protein SUMOylation on regulation for inflammatory response: To study the regulatory effect of protein SUMOylation in inflammatory diseases.
3) Protein SUMOylation and aging: To study the metabolic regulation effect of protein SUMOylation on aging.